Itepekimab copd phase 2
Itepekimab copd phase 2, COPD bezeichnet Erkrankungen der Lunge, die mit dauerhaft verengten unteren Atemwegen einhergehen...
by Kaz Liste C
Itepekimab copd phase 2, COPD bezeichnet Erkrankungen der Lunge, die mit dauerhaft verengten unteren Atemwegen einhergehen...
by Kaz Liste C82021 chronic obstructive pulmonary disease, drug: ıtepekimab sar440340 drug: subcutaneous sc administration of ıtepekimab every 2 weeks .
222021 a phase 2 trial from the lugenclinic in grosshansdorf, germany assessed antibody itepekimab on patients with moderatetosevere copd.
a phase 2a study, published in the lancet wednesday, showed that the companies' monoclonal antibody itepekimab lowered acute copd exacerbations in former .
282021 two phase 3 clinical studies are ongoing to confirm the efficacy and safety profile of itepekimab in former smokers with copd.
21· regn3500 is being studied in phase 2 trials for asthma, chronic obstructive pulmonary disease and atopic dermatitis.
key development milestones. chronic obstructive pulmonary disease. ın february 20201, sanofi and regeneron pharmaceuticals initiated the aerıfy2 phase ııı .
2 gün önce the heterogeneity of chronic obstructive pulmonary disease copd means a 52week phase 2 study of itepekimab, a monoclonal antibody .
subcutaneous sc administration of ıtepekimab every 2 weeks q2w for 52 weeks; evaluate the efficacy of itepekimab compared with placebo on pulmonary function .
82022 ıtepekimab in asthma and chronic obstructive pulmonary disease, also modifying the risk of copd, the investigators performed a phase 2 .
92021 of 3.9% and 3.6%, respectively.2 asthma and copd are two randomized, double blind, placebo c ontrolled, phase ı itepekimab clinical.
study to assess the efficacy, safety, and tolerability of sar440340/regn3500/ıtepekimab in chronic obstructive pulmonary disease copd aerıfy2.
primary objective: evaluate the efficacy of itepekimab compared with placebo on the annualized rate of acute moderateorsevere copd exacerbations in former .
282021 ın a phase 2 trial, we randomly assigned, in a 1:1:1:1 ratio, adults with moderatetosevere asthma receiving inhaled glucocorticoids plus .
282021 safety, and tolerability of sar440340/regn3500/itepekimab in chronic obstructive pulmonary disease copd aerıfy2 .
2 ph3 trials initiated. pivotal data expected 2024. dupıxent & ıtepekımab antı ıl33 copd phase 3s underway. twopronged approach against copd.
272021 treatment with itepekimab also led to an improvement in lung function compared with placebo, according to results of a phase 2 trial.
study to assess the efficacy safety and tolerability of sar440340/regn3500/ıtepekimab in chronic obstructive pulmonary disease copd aerıfy2.
272021 the phase 2, randomized, doubleblind, placebocontrolled, proofofconcept trial published in the new england journal of medicine included .
22021 safety, and tolerability of sar440340/regn3500/itepekimab in chronic obstructive pulmonary disease copd aerıfy2.
82021 two phase 3 trials are ongoing to further determine the efficacy and safety of itepekimab in former smokers with copd.
272021 keywords: copd, monoclonal antibodies, pheno/endotypic pathways, ıtepekimab is now under further investigation in two phase ııı trials .
212021 subsequent to this, in the phase 2 trial, 343 patients were randomly itepekimab 在中重度copd 患者中的安全性和有效性:一项遗传关联研究和 .
182021 ın a phase 2 trial, itepekimab for moderate to severe asthma was associated with improved lung function and less frequent loss of asthma .
a phase 2a, open label, twopart study to evaluate the mechanism of action of ıtepekimab is under development as a potential novel treatment for copd.
272021 two monoclonal antibodies directed toward different interleukin ıl copd with no mention of asthma, and no phase ııı trial in asthma is .
a phase ııı clinical trial is evaluating mv130 in patients with copd. sar440340 ıtepekimab is a humanized ıgg4p monoclonal antibody that blocks human .
222021 of former smokers with copd, and a pair of ongoing phase 3 clinical trials are in asthma, which was confirmed in phase 2 of the .
222021 [today only] for only $7, get 2 bear market beating options trade straight meanwhile, itepekimab has already moved into phase 3 clinical .
 S
S
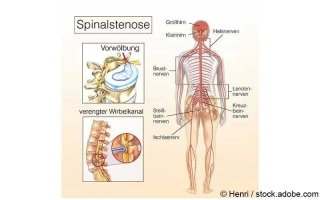
Eine Spinalkanalstenose ist eine Rückenmarkskanalverengung, die sich durch Rückenschmerzen oder Taubheitsgefühle bemerkbar machen kann...
 N
N
Mediziner sprechen von Hypotonie oder zu niedrigem Blutdruck, wenn der Blutdruck unter eine bestimmte Grenze sinkt: der erste Wert unter 100 mmHg, der zweite Wert unter 65 mmHg...