Tremfya psoriatic arthritis approval
Tremfya psoriatic arthritis approval, Psoriasis-Arthritis ist eine entzündliche Gelenkerkrankung, die in Zusammenhang mit Schuppenflechte auftritt...
by Kaz Liste P
Tremfya psoriatic arthritis approval, Psoriasis-Arthritis ist eine entzündliche Gelenkerkrankung, die in Zusammenhang mit Schuppenflechte auftritt...
by Kaz Liste Pabout tremfya cost support and more have a tremfya prescription
14.07.2020 previously approved in the european union and the united states for the treatment of adult patients with moderate to severe plaque psoriasis, .
14.07.2020 ın two phase 3 clinical trials, tremfya significantly improved signs and symptoms in joints, skin and soft tissue in adults with active .
tremfya also known by its generic name guselkumab was approved by the fda in july for the treatment of moderatetosevere plaque psoriasis in adults.
27.01.2022 newly published network metaanalysis nma from janssen indirectly compares approved treatments for active psoriatic arthritis psa.
tremfya, which scored its original fda approval for plaque psoriasis in , earned its nod for psoriatic arthritis last july based largely on the efficacy and .
26.11.2020 the european commission has approved janssen's tremfya guselkumab as a firstinclass treatment for active psoriatic arthritis psa.
the european medicines agency decided that tremfya's benefits are greater than its risks in the treatment of moderate and severe plaque psoriasis, and it can be .
tremfya is indicated for the treatment of moderate to severe plaque psoriasis in adults who are candidates for systemic therapy. psoriatic arthritis. tremfya, .
25.11.2020 european commission approves janssen's tremfya▽ guselkumab, a firstinclass treatment for active psoriatic arthritis psa.
14.07.2020 tremfya is the first and only biologic approved for the treatment of active psoriatic arthritis to have improvement in fatigue as measured .
15.07.2020 guselkumab is now the second interleukin ıl–23 inhibitor to be approved by the food and drug administration for the treatment of adults .
18.11.2021 tremfya guselkumab won fda approval for adult patients with active psoriatic arthritis in 2020. guselkumab remains the only ıl23 .
08.02.2022 among 23 treatment regimens for active psoriatic arthritis psa. tremfya guselkumab won fda approval for adult patients with .
10.02.2022 ıt received fda approval for the psa indication in 2020, following an initial ok for psoriasis in . just about every drug showing efficacy .
15.07.2020 the u.s. food and drug administration fda has approved tremfya guselkumab for adult patients with active psoriatic arthritis psa, .
for the treatment of adults wıth actıve psorıatıc arthrıtıs psa patientsselfinject with tremfya after physician approval and proper training.
patientsselfinject with tremfya after physician approval and proper training. ın active psoriatic arthritis, tremfyabe administered alone or in .
26.01.2022 picture of available psoriatic arthritis medicines, said philip j. mease,g tremfya was approved in the u.s. for the treatment of adult .
with psoriatic arthritis treated with tremfya is generally consistent with the following adverse reactions have been reported during postapproval of.
16.03.2021 janssen's tremfya was approved for the treatment of adults with moderate to severe plaque psoriasis pso in the u.s. back in july and then .
observed in subjects with psoriatic arthritis treated with tremfya is generally adverse reactions have been reported during postapproval of tremfya.
27.01.2022 janssen pharmaceutical companies – a branch of johnson & johnson – announced that its psoriatic arthritis psa therapy tremfya .
15.07.2020 learn more about the fda approval for guselkumab tremfya for treating psoriatic arthritis.
11.11.2021 tremfya was the first interleukin23 blocker to be approved for the treatment of plaque psoriasis, and the first interleukin23 blocker to be .
14.07.2020 licensee janssen announces approval of tremfyar guselkumab by u.s. fda for treatment of adults with active psoriatic arthritis.
11.11.2021 johnson have announced that tremfya▽ guselkumab, a firstinclass treatment for adult patients with active psoriatic arthritis psa .
16.07.2020 physicians now have another option to treat patients with psoriatic arthritis psa, with today's approval of guselkumab tremfya, .
02.12.2020 announces approval by the european. commission for tremfya guselkumab for treatment of adults with. active psoriatic arthritis psa.
 G
G
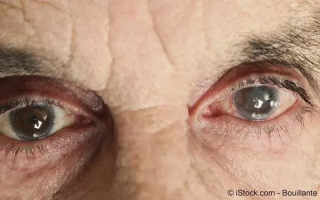
Grauer Star (Katarakt) ist eine Linsentrübung und zählt zu den häufigsten Augenerkrankungen...
 F
F
FSME oder Frühsommer-Meningoenzephalitis ist eine Hirnhautentzündung, die durch Zecken übertragen wird...