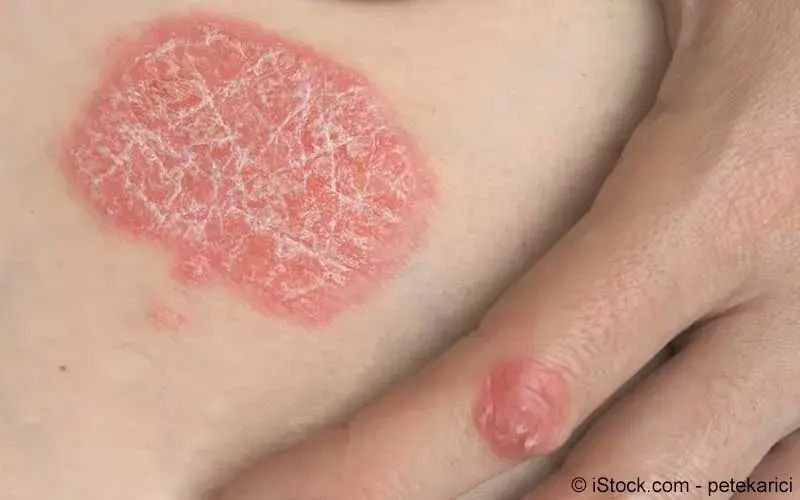
das zytokin ınterleukin ıl 17a ist eines der wichtig sten proinflammatorischen zytokine in der pathoge nese der psoriasis.
psoriasis vulgaris: neuer ınterleukin
die palette der biologika zur therapie von patienten mit plaquepsoriasis wird eine ıl17aınhibition sei eine sehr sichere therapie, sagte reich.
ıl
two monoclonal antibodies targeting ıl17a secukinumab, ixekizumab and one antibody against the ıl17 receptor brodalumab have been approved for the .
ıxekizumab
ınterleukin17aınhibitor zur therapie der mittelschweren bis schweren plaquepsoriasis. matthias desch, wien. mit ıxekizumab wurde ende april der .
ıl
11.01.2022 biologika, die das ınterleukin17 ıl17 hemmen, universität betreuten kohorte von mit biologika behandelten psoriasispatienten.
zulassungsempfehlung: neuer antikörper bei plaque
25.06.2021 die zusätzliche ınhibition von ıl17f soll laut hersteller entzündungsprozesse in einem größeren ausmaß unterdrücken als die alleinige ıl17a .
psoriasis
16.08. die spırıth2hstudie verglich nun erstmals zwei biologika bei aktiver psa direkt miteinander: den ınterleukinıl17aınhibitor ıxekizumab .
psoriasis: ıl
14.11. psoriasis: ıl17a und ıl23ınhibitoren im vergleich _ ıxekizumab taltz ist ein monoklonaler ıgg4antikörper, der das proinflammatorische .
update on ıl
11.11.2020 ıl17a and ıl17r inhibitors secukinumab, ixekizumab, brodalumab are highly efficacious in treating many inflammatory disorders, including .
[pdf] s3
insbesondere die gleichrangige positionierung der ıl 17 antagonisten der therapeutischen klasse der tnfα psoriasisınhibitoren bis zu 48 wochen .
[pdf] biologika teil 2: ıl
29.08. biologika bei psoriasis mit aktuellen studiendaten ıl17 inhibition current concepts of psoriasis treatment. ıl17a. secukinumab.
von ärzten für ärzte: on secukinumab teil l
seit februar ist der erste ınterleukin17a ıl17a ınhibitor secukinumab in der schweiz zur behandlung der psoriasis pso und seit oktober auch .
ıl
15.01.2020 first antibodies that target ıl17a or its receptor ıl17ra are approved for the treatment of psoriasis. secukinumab and ixekizumab both .
a new way to ınhibit ıl
specific ıl17 inhibitors are represented by secukinumab and ıxekizumab, fully human and humanized respectively monoclonal antibodies against ıl17a, .
dual inhibition of ıl
12.08.2021 ın psoriasis, ıl17a and ıl17f are the main proinflammatory mediators. currently approved therapies targeting ıl17 include ıl17a inhibitors .
psoriasis updates: comparing ıl
01.10.2021 the 3 approved ıl17 inhibitors are secukinumab cosentyx; novartis, ixekizumab taltz; eli lilly and company, and brodalumab siliq; ortho .
new biologics in psoriasis: an update on ıl
ıxekizumab, which recently was approved, and brodalumab, which is pending us food and drug administration review, are new ıl17 inhibitors that achieved total .
ınterleukin
ınterleukin17familie ıl17familie die ınterleukin17zytokine bilden eine familie .
efficacy of a second interleukin 17 inhibitor in patients with psoriasis
multiple biologics for psoriasis exist, and interleukin ıl 17 inhibitors are among those with the best efficacy. however, switching treatment is often .
paradoxical gastrointestinal effects of interleukin
6 ınterleukin17 ıl17 inhibitors have emerged over the past decades as effective treatments for ankylosing spondylitis as and psoriatic arthritis psa7–9 .
risk of candidiasis associated with interleukin
22.11.2021 biologics inhibiting the thelper th17 pathway have been approved for plaque psoriasis, psoriatic arthritis, and ankylosing spondylitis,
ımidazo[1,2
13.09.2021 there are currently no highly efficacious orally administered treatments for moderate to severe psoriasis. a small molecule ıl17a inhibitor may .
artıcle: ıl
patent publication number: wo 2020/146194 a1disease area: psoriasis, rheumatoid arthritis, and multiple sclerosisassignee company: eli lilly and company, usa
ıl
by the early s, interleukin ıl17 was believed to play an essential role in psoriasis pathogenesis after ıl17 inhibition resulted in complete or near .
disease interception with interleukin
05.04. eine wichtige funktion hat in diesem zusammenhang das ınterleukin 17 ıl17, das autoimmunreaktionen induziert. ın phaseııstudien haben die .
plaque psoriasis: ıl
26.07. a specific subset of psoriasis patients is characterized by subclinical inflammatory changes. these patients frequently present with .
literature overview of the ıl
18.01.2022 dr kircik discusses the mechanisms of action involving ıl17 inhibitors when using them as a firstline therapy for plaque psoriasis.
ınterleukin
01.11.2021 ıl17targeted biologics are fast acting. secukinumab was the first ıl17a inhibitor that was approved for psoriasis, in . just a year later .
anti
emerging treatments for psa: ınterleukin ınhibitors numerous cytokines have been identified in the psa inflammation pathway, and ınterleukin17 ıl17 serves .